Reduced Expression of MTSS1 Increases Sarcomere Number and Improves Contractility in Select Forms of Monogenic DCM
Authors: Hannah Kleppe, Anastasia Budan, Luke Zhang, Marie Majetic, Reva Shenwai, Alan R. Levinson, Olga Cisne-Thompson, Bernardo Zepeda, Charles E. MacKay, Farshad Farshidfar, Jonathan H. Tsui, Sylwia M. Figarska, Timothy Hoey, James R. Priest, and Rebecca E. Slater
Originally Published in: JACC: Basic to Translational Science (October 2025) (Download)
Summary
Beyond the Mutation: How Human Genetics and 3D EHTs Uncovered a New Therapeutic Target for DCM🫀
Dilated Cardiomyopathy (DCM) affects up to 1 in 250 adults and remains a leading cause of heart failure characterized by reduced cardiac output and ventricular thinning. While genetic factors contribute to 50% of cases where the titin (TTN) locus is the most common, there are currently no approved therapies that directly address the underlying contractile decay and sarcomere dysfunction.
MTSS1 as a Genetic Protective Factor
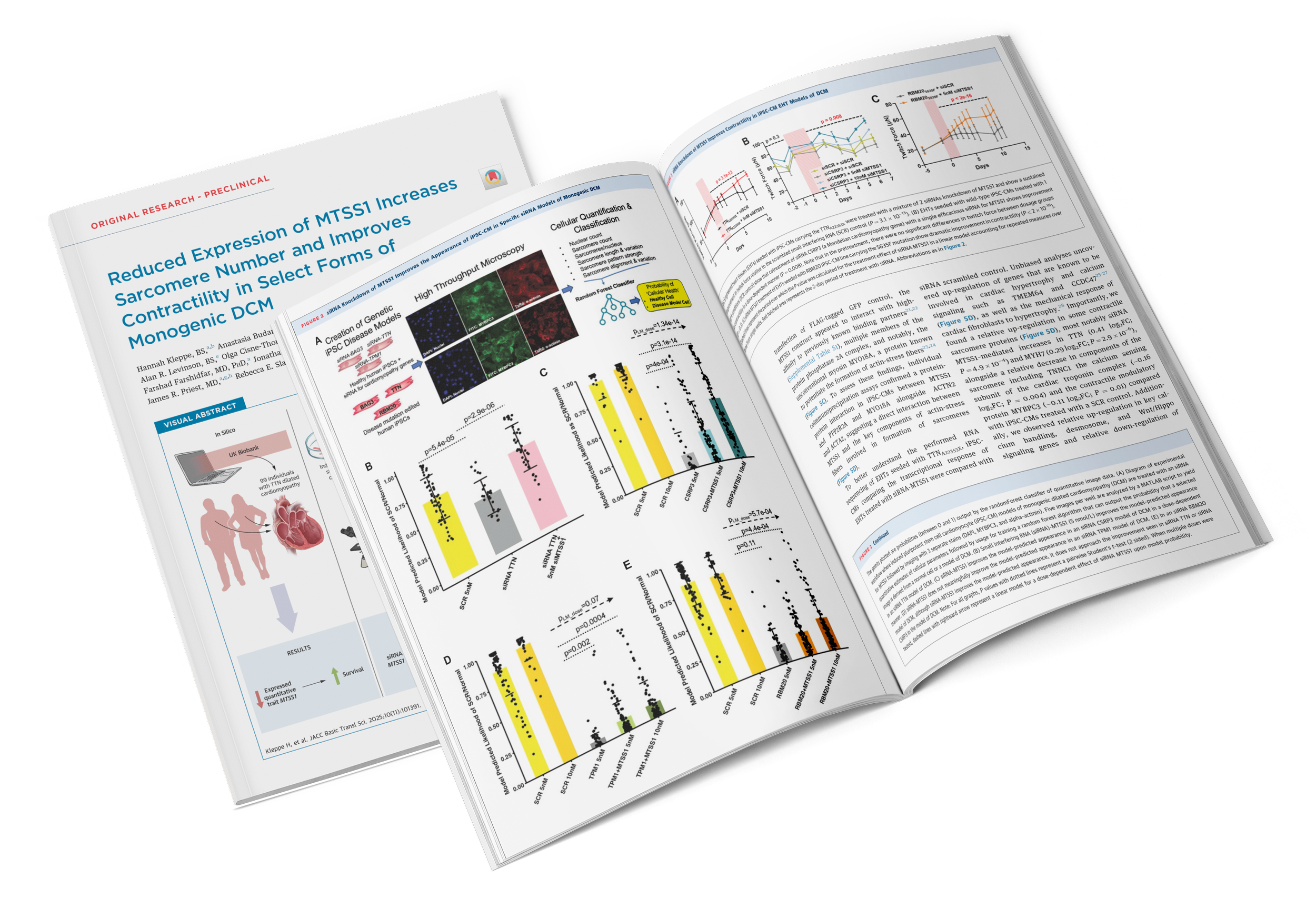
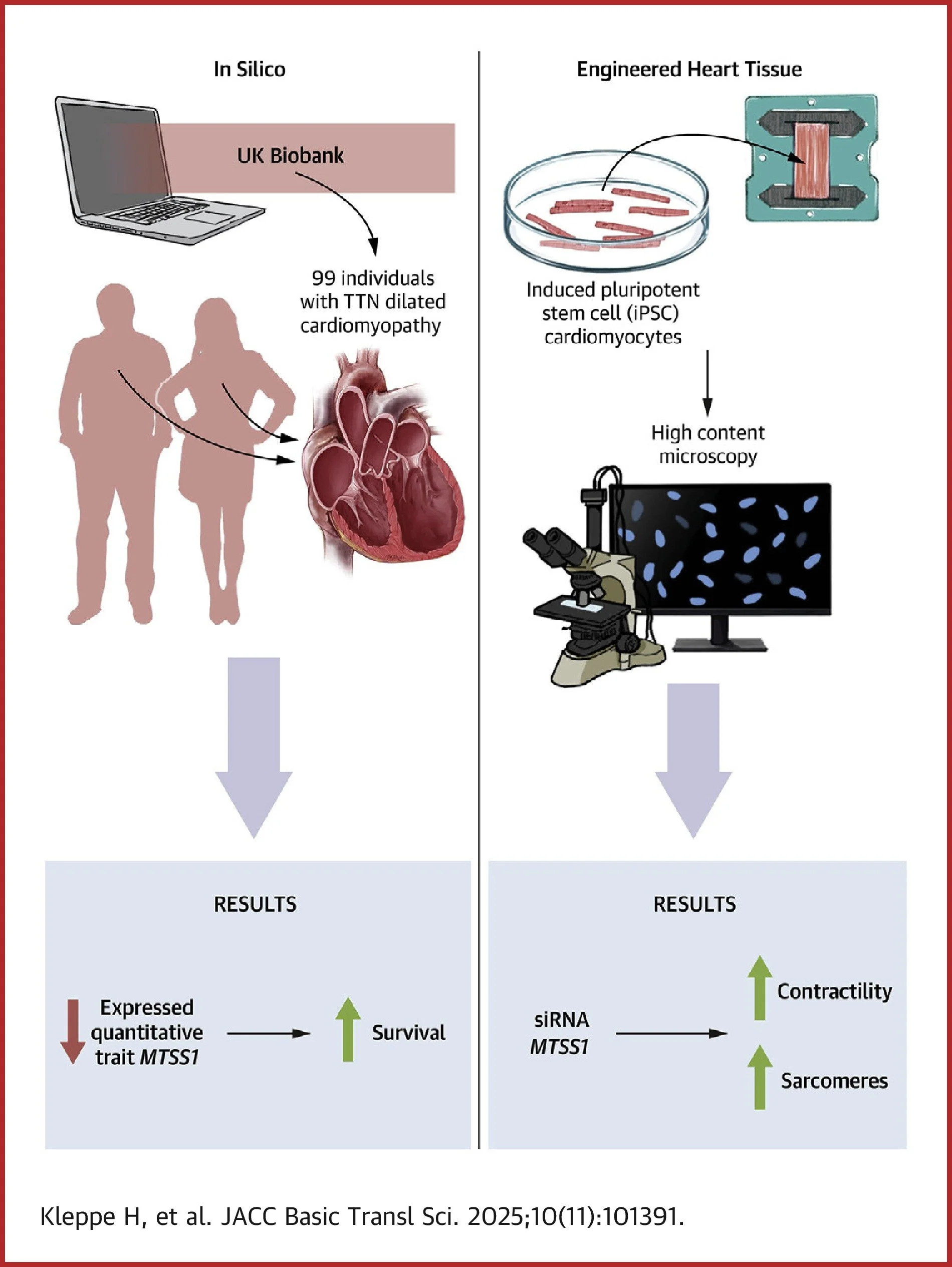
A recent study published in JACC: Basic to Translational Science has uncovered a "roadmap" for heart failure drug discovery. By analyzing the UK Biobank, the team from Tenaya Therapeutics identified 99 individuals with pathogenic TTN mutations who developed DCM. They found that those who also carried a specific genetic variant (rs12541595) that naturally lowers the expression of the protein MTSS1 in the left ventricle had significantly improved survival rates.
This human genetic data suggested a powerful hypothesis: if we can pharmacologically or genetically lower MTSS1 levels, can we "rescue" the failing heart?
Building the 3D Human Heart in the Lab
To validate these findings, researchers moved from "in silico" data to "in vitro" human models using Curi Bio’s 3D Engineered Heart Tissue (EHT) ecosystem. Unlike traditional 2D monolayers, which lack the mechanical cues of native tissue, these 3D EHTs replicate the physical environment of the human heart. The study used human iPSC-derived cardiomyocytes across multiple genetic backgrounds (TTN, CSRP3/MLP, and RBM20) to demonstrate the broad potential of MTSS1 knockdown.
Key findings using Mantarray™ and other platforms:
Sarcomere Restoration: siRNA knockdown of MTSS1 led to a reproducible increase in the total number of sarcomeres per cardiomyocyte.
Significant Twitch Force Improvement: In TTN mutant models, MTSS1 knockdown resulted in a 147% improvement in twitch force.
Broad Therapeutic Applicability: Beneficial effects were also seen in CSRP3 and RBM20 backgrounds, with the RBM20 models showing a dramatic 284% improvement in contractility.
Integrated Molecular Insight: Using mass spectrometry and RNA sequencing, the team discovered that MTSS1 acts as a negative regulator of sarcomere formation by interacting with MYO18A, a protein critical for sarcomere assembly.
Translational Impact: De-risking the Pipeline
The study also highlighted a common pitfall in drug development: the failure of animal models to translate. When researchers attempted to induce these same DCM phenotypes in mouse models using various stressors, they were unable to reproduce the cardiac dysfunction seen in humans.
This reinforces why the Curi Bio 3D EHT ecosystem is vital for modern pharma. By providing human-relevant, quantitative readouts of cardiac structural and functional decay, we enable researchers to validate targets based on human biology before moving to clinical trials.
This #AmericanHeartMonth, we celebrate this step toward precision medicine. By linking human genetics with predictive 3D human models, we are unlocking the biology needed to solve the heart’s most complex pathologies.