Mantarray™ App Notes
Machine Learning-Guided Exercise Protocol Development – An Adaptable Workflow for Stimulation Parameter Identification in 3D NMJs
Featuring: Mantarray Platform | Neuromuscular Junction Model (Released March 2026)
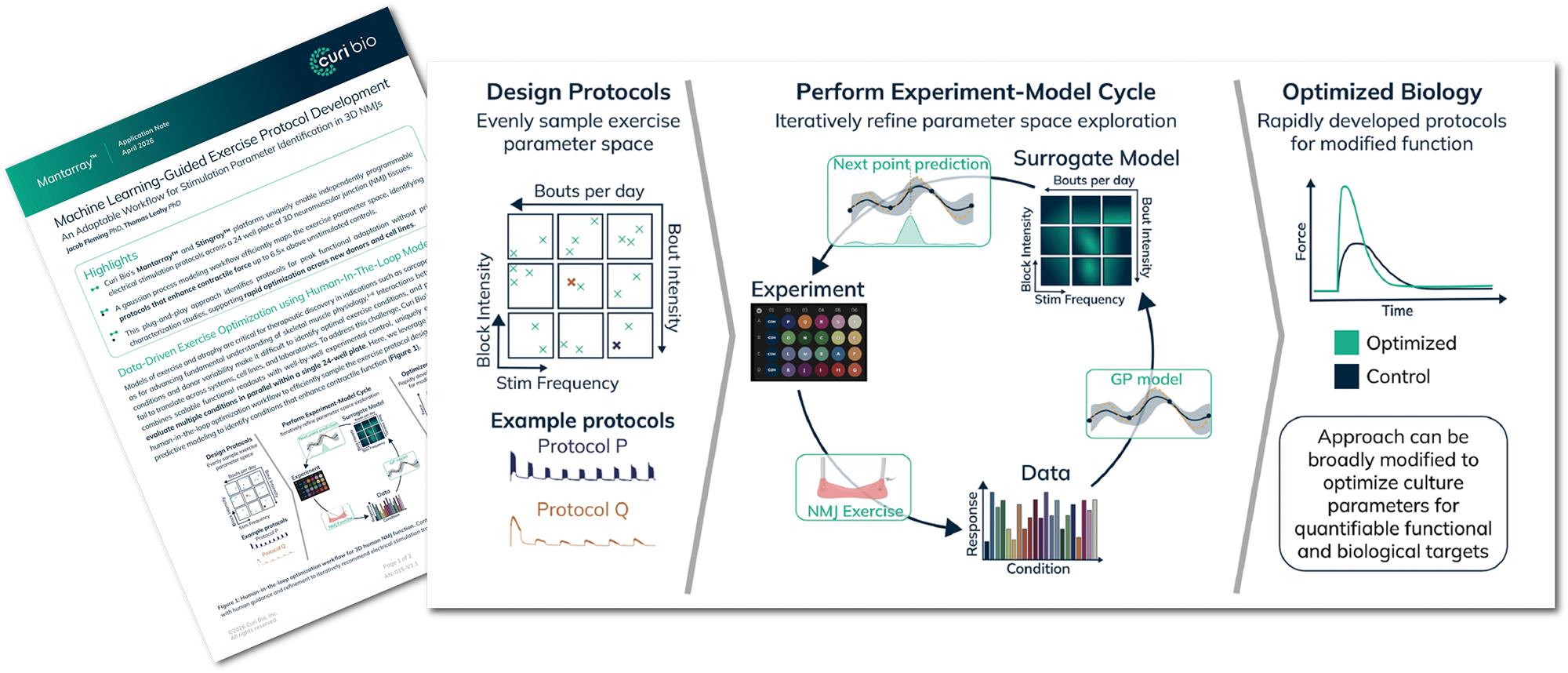
Massive Functional Gains: Optimized stimulation protocols drive superior tissue maturation, enhancing contractile force up to 6.5x above unstimulated controls.
Exercise Protocol Parameter Mapping: Characterize hypertrophic adaptation through multi-dimensional analysis of stimulation frequency, block intensity, and daily bout density.
High-Throughput Iteration: Eliminate inter-experiment variability and reduce costs by executing complex Design of Experiments (DoE) within a single, high-throughput plate.
Predictive Validation: Use leave-one-out cross-validation to assess model performance and establish standardized, cell-line-specific parameters.
Broadening Motor Neuron Sources for 3D Human NMJs – Rapid Engineering of 3D Human NMJs for Scalable Disease Modeling Applications
Featuring: Mantarray Platform | Neuromuscular Junction Model (Released March 2026)
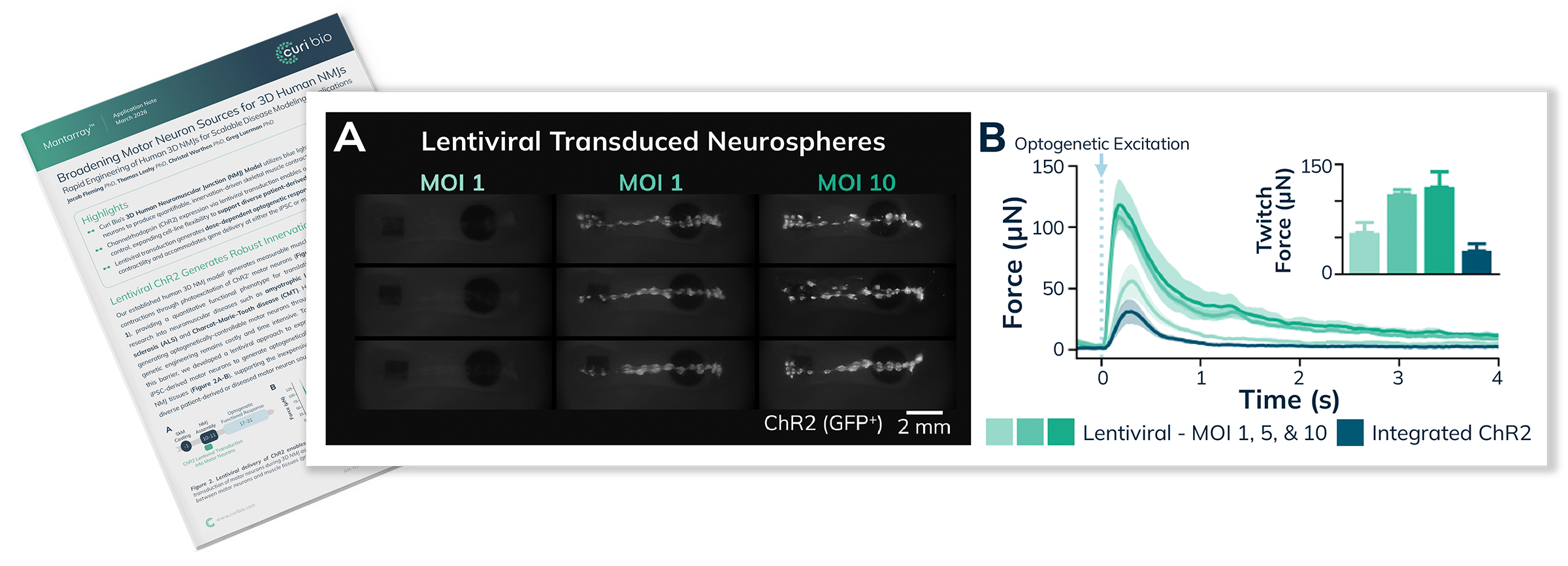
Curi Bio’s 3D Human Neuromuscular Junction (NMJ) model utilizes blue light stimulation of motor neurons to produce quantifiable, innervation-driven skeletal muscle contractions.
Channelrhodopsin (ChR2) expression produces dose-dependent optogenetic responses while preserving muscle contractility, with ChR2 delivery supported at the iPSC or motor neuron stage.
Lentiviral transduction of ChR2 enables efficient engineering of optogenetic control, expanding compatibility with diverse motor neuron sources for translational neuromuscular disease modeling.
Functional Potency Testing of Botulinum Neurotoxins – A Scalable 3D Human NMJ Model to Replace the Mouse Lethality Bioassay
Featuring: Mantarray Platform | Neuromuscular Junction Model (Released March 2026)
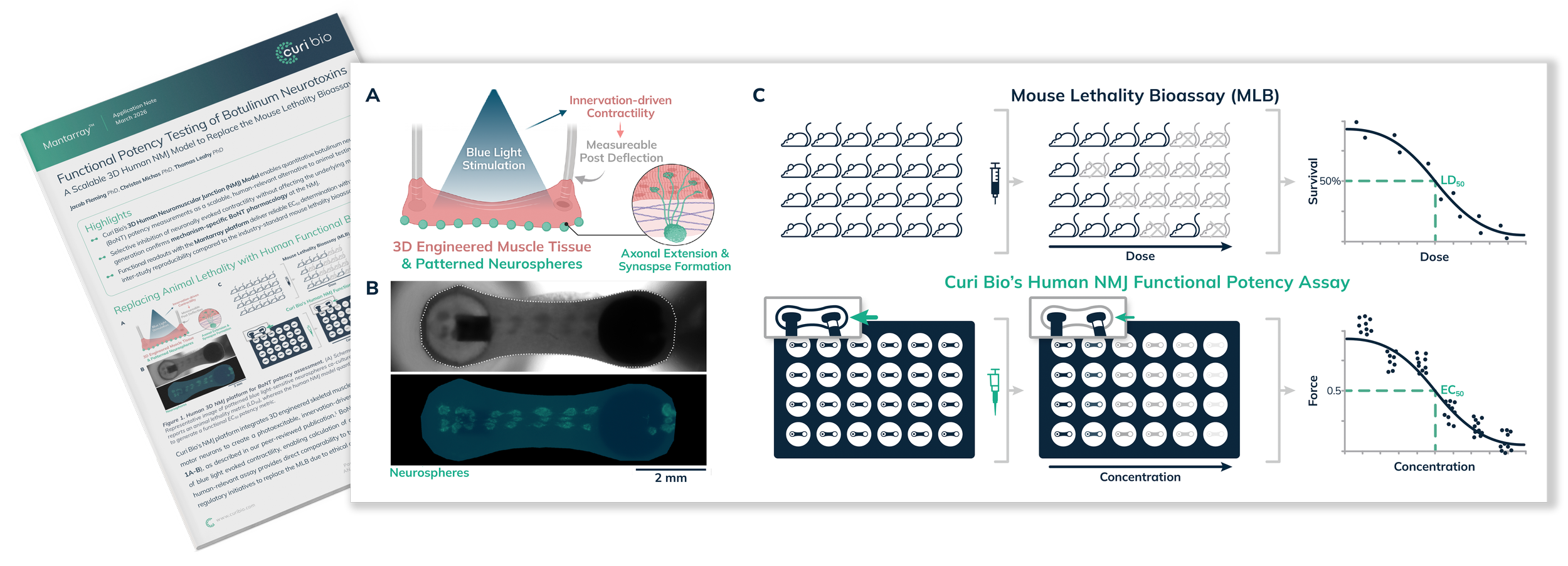
Curi Bio’s 3D Human Neuromuscular Junction (NMJ) Model enables quantitative botulinum neurotoxin (BoNT) potency measurements as a scalable, human-relevant alternative to animal testing.
Selective inhibition of neuronally evoked contractility without affecting the underlying muscle force generation confirms mechanism-specific BoNT pharmacology at the NMJ.
Functional readouts with the Mantarray platform deliver reliable EC50 determination with >10x improved inter-study reproducibility compared to the industry-standard mouse lethality bioassay (MLB).
Modeling Human Cardiac Arrhythmia in 3D – Rapid Disease Model Development via siRNA-Mediated PKP2 Knockdown
Featuring: Mantarray Platform | Pulse Analysis Software (Released February 2026)
The Mantarray platform enables disease-relevant functional measurements in 3D engineered heart tissues (EHTs), including altered contractility and arrhythmic behavior.
siRNA-mediated knockdown of PKP2 provides a rapid, non-genetic strategy to model desmosomal arrhythmia in human EHTs, supporting flexible and scalable disease modeling.
Pulse analysis software enables robust analysis of 3D contractility and fluorescence data, with adaptable parameters to accurately identify arrhythmic patterns in irregular force traces.
3D Skeletal Muscle Modeling with Primary Human Cells – Applications in Metabolic Regulation and Muscle Atrophy
Featuring: Mantarray Platform | Cook MyoSite’s Single-donor Human skMDCs (Released January 2026)
Primary human skeletal muscle-derived cells (skMDCs) from Cook MyoSite enable integrated functional and metabolic profiling when cultured as 3D engineered muscle tissues (EMTs).
EMTs exhibit insulin sensitivity and inducible atrophy, supporting human-relevant modeling for emerging clinical challenges such as GLP-1 receptor agonist-associated lean muscle loss.
Positive Force-Frequency Relationship in iPSC-derived Engineered Heart Tissues
Featuring: Mantarray Platform | Curi Bio Cardiac Media (Released May 2025)
Positive force-frequency relationship (FFR) achieved in iPSC-derived heart tissues using Mantarray™ and continuous electrical stimulation with Curi Bio’s Cardiac Media.
Maximal force observed at 2.75 Hz by day 22, indicating advanced cardiac maturation.
Stimulated tissues showed 43% higher force and 23% faster relaxation vs. controls.
Unstimulated controls had negative FFR, despite higher baseline force.
FFR gains reversed after stimulation stopped; ongoing work explores metabolic cost and protocol optimization.
Functional Atrophy Induced by Dexamethasone in Primary and iPSC-derived Engineered Skeletal Muscle Tissues
Featuring: Mantarray Platform | iPSC-derived Skeletal Muscle Myoblasts | Neuromuscular Junction Model (Released February 2025)
Dexamethasone induces dose-dependent, reproducible, and reversible reductions in force generation in engineered skeletal muscle tissues (EMTs).
Both iPSC-derived and primary human EMTs showed significant atrophy, with primary cells losing all measurable contractile force at high doses.
Recovery after dexamethasone removal was slow and incomplete over a 21-day period.
The Mantarray Platform provides a robust model for studying atrophic signaling and testing potential therapeutic strategies.
A Scalable Functional Model of 3D Human Neuromuscular Junctions Using the Mantarray Platform
Featuring: Mantarray Platform | iPSC-derived Skeletal Muscle Myoblasts | Neuromuscular Junction Model (Released February 2024)
Facile co-culture of iPSC-derived neurons & skeletal muscle into a 3D NMJ model.
Scalable, reproducible & reliable formation of 3D human NMJs with long in vitro lifetimes.
Turnkey, longitudinal collection of NMJ functional data, backed by histological evidence.
Pre-clinical Identification of Dose-dependent Cardiac Toxicity
Featuring: Mantarray Platform (Released January 2024)
Turnkey scalable production of human 3D engineered heart tissues.
Label-free automated capture and analysis of functional data across 24 tissues in parallel.
In vitro validation of clinical drug toxicity and efficacy using human cell models.
Identification of optimal dosing regimens to minimize functional side-effects.