Assay-Ready iPSC-Derived
DM1 Skeletal Muscle Myoblasts
Patient-specific human myoblasts and CRISPR-corrected isogenic control — optimized for 3D engineered muscle tissue research and therapeutic discovery
| iPSC-Skeletal Muscle Line | Genetic Mutation | Clinical Disease | Price |
|---|---|---|---|
|
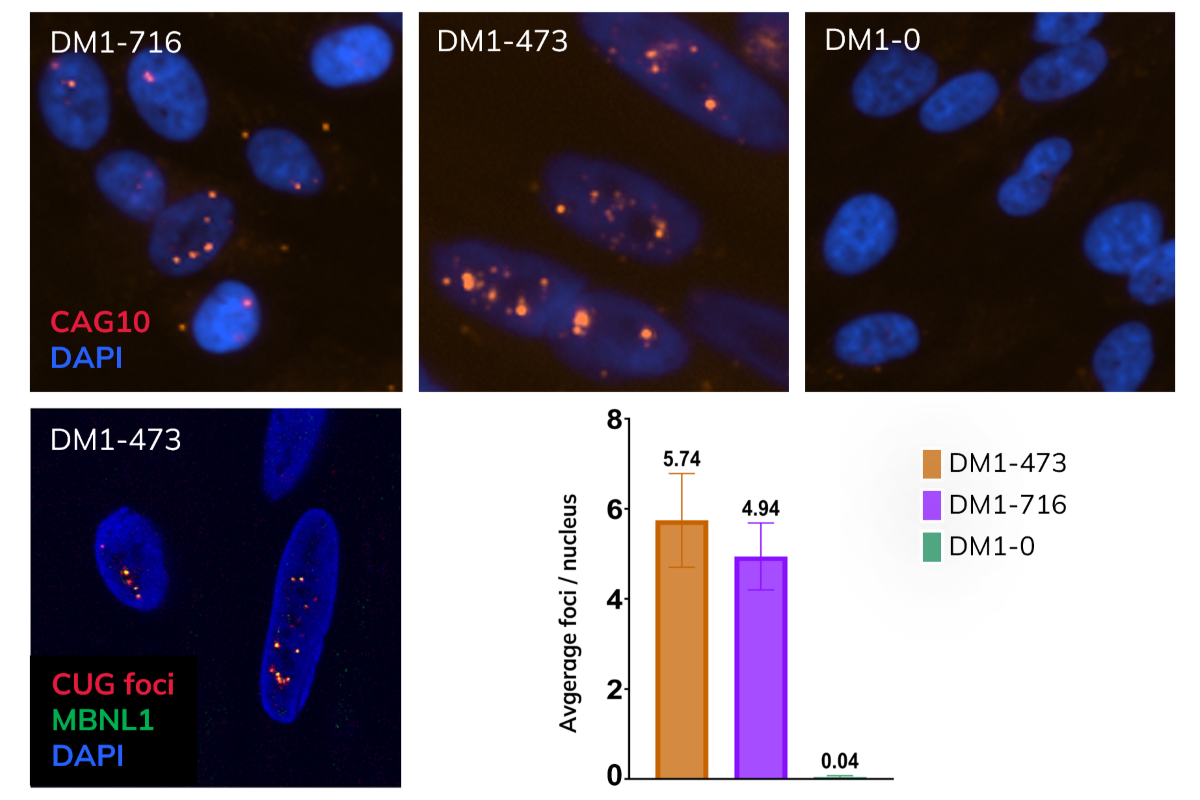
DM1-716 (Disease) 35 y.o. male |
CTG trinucleotide repeat expansion in 3′ UTR of DMPK 716 CTG repeats (~2,100 nt) |
Myotonic Dystrophy Type 1 | $3,295 6 million cells $5,995 12 million cells *10 day supply of media included |
|
DM1-473 (Disease) 18 y.o. male |
CTG trinucleotide repeat expansion in 3′ UTR of DMPK 473 CTG repeats (~1,500 nt) |
Myotonic Dystrophy Type 1 | |
|
DM1-0 (Isogenic Control) |
0 CTG repeats (CRISPR-corrected) · WGS confirmed | Isogenic control to DM1-716 | |
|
24-WT (Unrelated Wild-Type) |
Normal CTG repeat length | Healthy reference control | $2,550 6 million cells $4,750 12 million cells *10 day supply of media included |